2023 Q1 Update – Message from the CEO
Hello friends, advisors, and collaborators.
We've come a long way in a year. Your support and encouragement has made it possible to get to this point. Some highlights:
Filed provisional and nonprovisional patent applications for a small-joint implant device
Validated $4.2B market, regulatory pathway, reimbursement, and competitive landscape through intense commercialization research with NSF I-Corps and LifeX life sciences accelerators
Awarded $20,000 first prize from LifeX Labs pitch competition
Learned a ton about grants and grant writing
Recruited a powerful informal advisory board and engineering team ready to go on R&D
Now we need to get into the lab and validate the technology. We are looking for $50k - $125k, ideally in non-dilutive funding through economic development and foundation grants or competitions plus a small private raise to bridge timing gaps. Let me know if you are interested in learning more about this pre-seed round that will offer a risk premium.
Honor Roll
So many people have helped make this quest a reality. When I found out after my table saw accident that joint replacement for advanced arthritis in fingers is rare and risky due to deficiencies in current technology, I got motivated. My excellent hand surgeon, Dr. Ryan Katz at the Curtis National Hand Center in Baltimore, was equally inspired so we partnered up to address this huge unmet need.
Thank you, thank you, THANK YOU to all who have contributed. We would not be this far along without you, including all the surgeons we interviewed and many others not listed below.
Allan C. Doyle
Deb Hemingway
Andrew Abadeer, MD
Nicholas Calotta, MD
Grady White, Esq.
Abba Poliakoff, Esq.
NSF I-Corps UMD team
LifeX Accelerator cohort
Kelly Collier
Laura Ohlund
Glenn Watson
Michele Miguiolo
Julie Gulick
Rishaan Sharma
David H. Gibel
Sadaf Akbari
Alexandre Cavalca
Hugh Bruck, Ph.D.
Cyrus Etemad-Moghadam
Ben McCandless
Billy Petzold
Ed Rothstein
... and many more
Looking back...
It's encouraging to be reminded of how much can be accomplished with hard work and great support. [image]
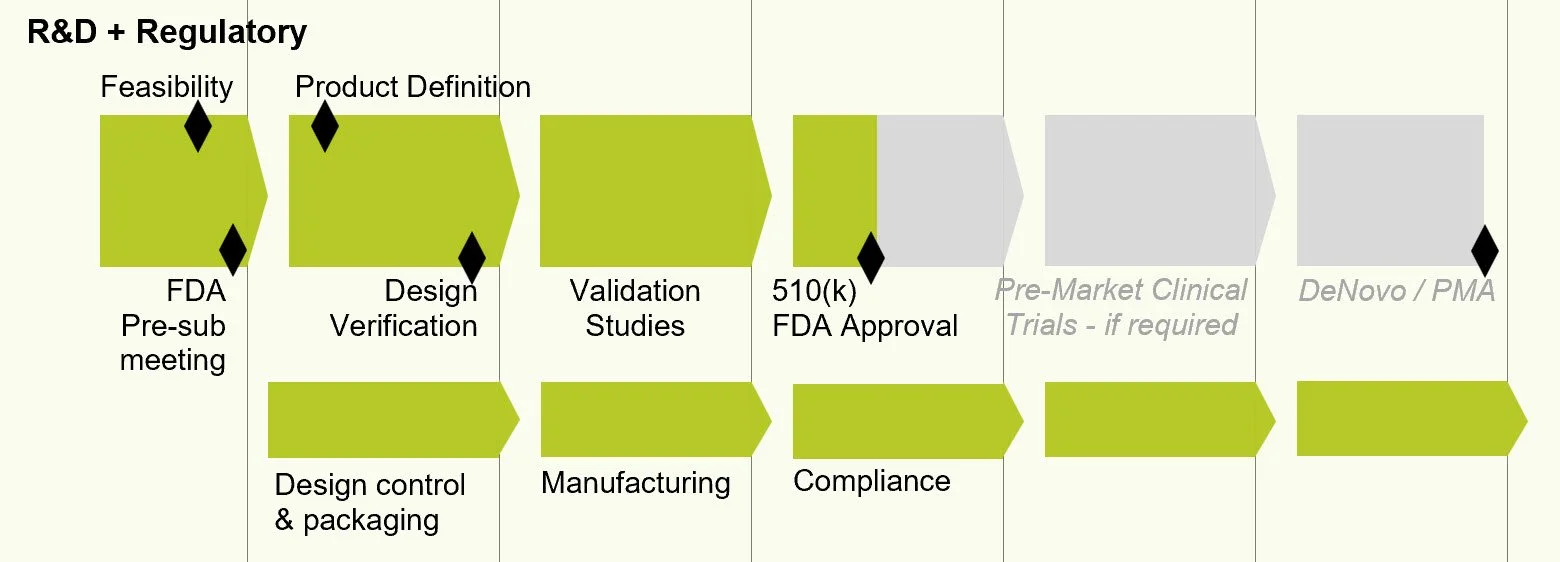
The work ahead
The years ahead are all about R&D and regulatory compliance for FDA approval. While business operations demand attention, critical activity will be in the lab. Grant writing and fundraising goes with the territory. We'll expand the team along the way. If you know of people who might want to join us, I would welcome your introduction. We're going to need grant writing, R&D, regulatory, business planning and market expertise. [image]
That's it for now. Please contact me with any questions or comments. I'd be happy to share more detail with you. My inbox is open for ideas, investment queries, introductions, and general encouragement.
Thanks, everyone. Let's stay in touch.
Gratefully,
Marcia Hart, CEO
mobile: 667-444-9322
Djit Medtech, LLC